Traci Jesse Stevens | 2000-2004
 Postdoctoral Fellow
Postdoctoral Fellow
Ph.D. Washington University
Funded by an NIH Postdoctoral Fellowship
Currently an Associate. Professor at Randolph-Macon College
The effects of activating Abelson kinase: A model for Bcr-Abl function in leukemia
Adherens junctions are multiprotein complexes that mediate cell-cell adhesion and are critical for tissue organization and morphogenesis. The adhesive function of adherens junctions is dependent on cadherins. Drosophila Armadillo (Arm), the homologue of vertebrate ß-catenin, is a key component of adherens junctions that mediates linkage of cadherins to the actin cytoskeleton and is critical for cell-cell adhesion during development. We are interested in understanding how actin polymerization and contractility is regulated at adherens junctions in epithelial cells.
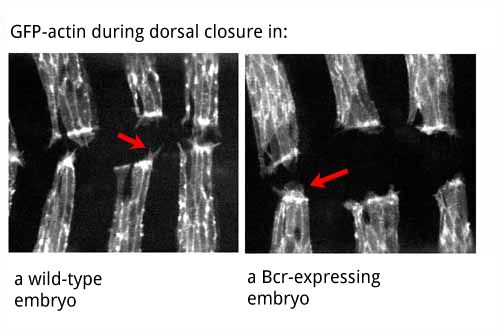
Studies in our lab indicate that the non-receptor tyrosine kinase Abelson (Abl) and its substrate, Enabled (Ena), play a role in cytoskeletal regulation during cell-cell adhesion. Ena regulates actin dynamics and cell motility of fibroblasts. To provide insight into the functions of Abl kinase during cell adhesion, we are assessing the effects of increasing Abl tyrosine kinase activity, by characterizing the phenotypes of embryos ubiquitously expressing an oncogenic, activated form of Abl, Bcr-Abl. The Bcr-Abl fusion protein results from a chromosomal translocation and plays a causative role in certain human leukemias. In cell culture, expression of Bcr-Abl results in increased cell motility and rearrangements of the actin cytoskeleton.
We have found that Drosophila embryos ubiquitously expressing Bcr-Abl die with defects in morphogenetic processes that require cell shape changes and cell migration, including dorsal closure, head involution, and germband retraction. Similar defects are observed in embryos over-expressing wild-type Abl, while embryos over-expressing mutant forms of Bcr-Abl or Abl that abolish tyrosine kinase activity are viable and have no defects in embryonic development. These results support the idea that regulated tyrosine kinase activity of Abl is critical for cell-cell adhesion in epithelial tissues. We hypothesize that Abl substrates may play key roles at adherens junctions and that Abl phosphorylation regulates their function. In support of this, Bcr-Abl localizes primarily to cell junctions; similarly, overall levels of tyrosine phosphorylation are increased at cell junctions. Furthermore, we have observed increased tyrosine phosphorylation of Ena and other Ena-binding proteins in the presence of Bcr-Abl. Expression of Bcr-Abl during embryonic development results in the loss of Ena localization to the leading edge and to cell junctions. This is consistent with parallel studies in our lab on abl mutants that suggest that Abl negatively regulates Ena localized to certain sites, thus influencing actin polymerization. Our preliminary studies also suggest that activating Abl disrupts the normal coordinated rearrangements of the actin cytoskeleton that occur during dorsal closure. Studies are currently underway to examine the dynamic behavior of the actin cytoskeleton in live embryos expressing Bcr-Abl.
Publications
- Jesse TL, LaChance R, Iademarco MF, Dean DC. (1998) Interferon regulatory factor-2 is a transcriptional activator in muscle where It regulates expression of vascular cell adhesion molecule-1. Journal of Cell Biology 140, 1265-76
- Grevengoed, E.E., Loureiro, J.J., Jesse, T.L., and Peifer, M. (2001). Abelson kinase regulates epithelial morphogenesis in Drosophila . Journal of Cell Biology 155, 1185-1197.
- Stevens, T L., Rogers, E M., Koontz, L. M. , Fox, D. T., Homem, C. C.F., Nowotarski, S. H., Artabazon, N. B., and Peifer, M. (2007). Using Bcr-Abl to examine mechanisms by which Abl kinase regulates morphogenesis in Drosophila. Molecular Biology of the Cell.


