Noah Gurley
Background & Contact Information
Position Post-Baccalaureate Research Assistant (2021 – Present)
Education: B.Sc. in Quantitative Biology at UNC-CH (w/ a minor in Neuroscience) 2021
Email: ngurley@unc.edu
Research Information
Cadherin-based adherens junctions (AJs) promote coordinated behavior in epithelial cells by facilitating a connection between the apical membranes and actomyosin cytoskeletons of neighboring cells. The proper assembly, positioning, and function of AJs requires assembly of a large multiprotein machine at the apicolateral membrane, via interactions between specific domains of these proteins.
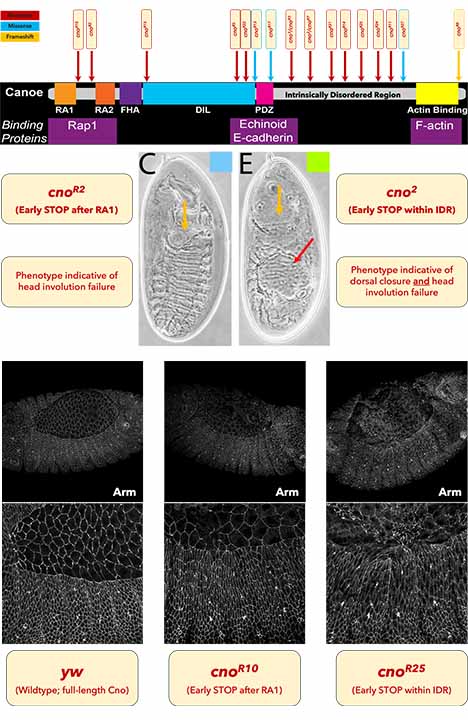
Canoe (Cno) is a large scaffolding protein and is an orthologue of the mammalian protein Afadin. The full-length, wild-type Cno protein sequence contains multiple structural domains, three of which have been of particular interest to our lab in recent years; these include the Ras-associated, PDZ-binding, and filamentous actin-binding domains. Specific domains of Cno interact with junctional, cytoskeletal, and regulatory proteins; however, the specific contribution of many domains to Cno’s function remains largely uncharacterized in vivo. Additionally, our lab is interested in interrogating the function of Cno’s intrinsically disordered region using “classical” alleles, which were generated via EMS mutagenesis.
Currently, my research in the Peifer Lab is centered around characterizing the specific functional contribution of Cno’s various protein domains. cno2 is the original allele from which the canoe phenotype got its name; it contains a nonsense mutation early within the intrinsically disordered region. Conversely, the canonical “null” allele – cnoR2 – has an early nonsense mutation after the first RA domain. Intriguingly, despite having early truncation that prevents the production of a detectable protein product, cnoR2 exhibits a less severe cuticle phenotype compared to cno2. The more severe phenotype of cno2 suggests that cno2 may produce a truncated protein that negatively interact with the maternally contributed wildtype Cno.
To further investigate this possibility, we have characterized an additional 20 cno alleles that (primarily) contain nonsense mutations at various locations within the coding sequence. In working my way through these mutants, I am performing for the following for each allele:
- Assessing the effects on morphogenesis of zygotic mutants via analysis of cuticle phenotypes
- Assessing embryonic phenotypes of zygotic mutants at the cellular level via immunostaining and confocal imaging – during dorsal closure, a major morphogenetic process in which the cells of two epidermal sheets experience a dramatic increase in tension as the two lateral sheets dorsally converge, and subsequently fuse, to close an epithelial gap
- Analyzing cuticle phenotypes of germline clones to assess the influence of maternal contribution of wildtype protein and to parse out the potentiality of antimorphic interactions between mutant and wildtype protein products
Publications
Gurley, N.J., and Peifer, M. (2024). Moonwalking Molecular Machines: Unraveling the Choreography of Myosin Filament Assembly. Journal of Cell Biology, in press.
McParland, E.D.*, Butcher, T.A.*, Gurley, N.J., Johnson, R.I., Slep, K.C., and Peifer, M. (2024) Canoe’s Dilute domain is not essential for linking cell junctions to the cytoskeleton but supports morphogenesis robustness. Journal of Cell Science, in press. *=co-first authors.
McParland, E.D.*, Butcher, T.A.*, Gurley, N.J., Johnson, R.I., Slep, K.C., and Peifer, M. (2024) Canoe’s Dilute domain is not essential for linking cell junctions to the cytoskeleton but supports morphogenesis robustness. Journal of Cell Science, in press. *=co-first authors.
Gurley, N.J., Szymanski, R.A., Dowen, R.H., Butcher, T.A., Ishiyama, N. and Peifer, M. (2023). Exploring the evolution and function of Canoe’s intrinsically disordered region in linking cell-cell junctions to the cytoskeleton during embryonic morphogenesis. PLoS One 18: e0289224.
, , , ,