John Poulton Alumna Page
Background & Contact Information
Currently: Research Assistant Professor of Medicine, UNC-CH and UNC’s Kidney Center
Postdoctoral Fellow in Peifer lab (2009- 2016)
Education: PhD, Florida State University, Tallahassee – Department of Biology
Fellowships: American Heart Association Predoctoral Fellowship
NIH Postdoctoral Training Grant, Lineberger Comprehensive Cancer Center
NIH NRSA Individual Postdoctoral Fellowship
Email: poultonj@email.unc.edu
DOWNLOAD CV
Research Information
Cell division is a tightly regulated process, ensuring that accurate chromosome segregation, via microtubule-based mitotic spindles, occurs with high fidelity. The cytoskeleton is critical for mitosis, facilitating spindle formation and chromosome segregation. Defects in spindle formation or function can lead to chromosome mis-segregation and aneuploidy, a common form of chromosomal instability (CIN) and hallmark of most cancer cells. Centrosomes are the primary microtubule organizing centers of animal cells and are key contributors to mitotic spindle assembly. To ensure mitotic fidelity, checkpoint pathways (e.g. the DNA damage response and Spindle Assembly Checkpoint) monitor for specific defects that may occur, either due to random errors or as a result of mutations in genes that promote the fidelity of the process. In cancer, cells lose their ability to control the cell cycle, but many tumors also possess additional mutations in cellular checkpoints and exhibit centrosome dysfunction, which are believed to contribute to genomic instability and disease progression. My research is focused on characterizing the relative contributions of, and interplay between, cytoskeletal regulators and cellular checkpoints during mitosis, with particular interest in uncovering how defects in these processes can lead to chromosomal instability and oncogenesis.
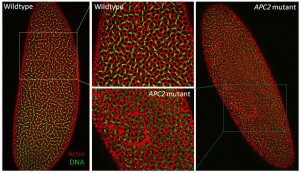
I began my postdoctoral research investigating the cytoskeletal roles of the tumor suppressor Adenomatous Polyposis Coli (APC) in mitosis, using the early fly embryo as a model system. APC mutations are the initiating mutation in ~80% of all cases of colorectal cancer, and play roles in both tumor initiation, via their effect on Wnt signaling, and tumor progression, via their roles in cytoskeletal regulation. Our studies revealed that APC promotes accurate chromosome segregation through its role in centrosome separation, and also suggested that cells have backup mechanisms that compensate for errors resulting from the absence of APC (Poulton et al., Development,2013).
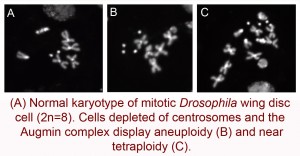
These experiments stimulated my interest in how cells respond to the loss of mitotitc fidelity regulators more generally, and I turned my attention to centrosome function. Many tumors show misregulated centrosome number or function, suggesting centrosomes serve a central role in preventing CIN and cancer. Mutations in centrosomal proteins also underlie primary microcephaly (MCPH), a developmental disorder resulting in reduced brain size. However, in both cancer and MCPH, it remains unclear how defects in centrosome function contribute to disease, underscoring the need for in vivo, mechanistic examinations of centrosomes in mitosis and development. While the textbook view suggested that centrosomes are the primary microtubule nucleating centers of animal cells, with key roles in forming and orienting mitotic spindles, the survival of fruit flies to adulthood in the absence of centrosomes suggested they are dispensable in somatic cells, challenging the canonical view. I used the fly wing disc as a model epithelium to resolve these conflicting hypotheses. We found that centrosomes do play vital roles, as they are essential for efficient spindle assembly, accurate chromosome segregation, and proper spindle orientation within the tissue. In the absence of centrosomes, defects in these processes activate the JNK signaling pathway, which triggers cell death, disrupting proper tissue development. We also found that multiple mechanisms buffer the effects of centrosome loss, including alternative microtubule nucleation pathways and the Spindle Assembly Checkpoint (Poulton et al., Developmental Cell, 2014). I am continuing to explore how centrosomes work in concert with other microtubule nucleation pathways and cellular checkpoints to promote mitotic fidelity, cell viability, and tissue development, both in the epithelia of the wing disc and the neural stem cells of the developing brain, as a model of microcephaly.
Publications
- Poulton J and A Winn. (2002) Costs of canalization and plasticity in response to neighbors in Brassica rapa. Plant Species Biology. 17:109-118
- Poulton J and W-M Deng. (2006) Dystroglycan down-regulation links EGF receptor signaling and anterior-posterior polarity formation in the Drosophila oocyte. Proceedings of the National Academy of Sciences USA. 103:12775-12780
- Schneider M, Khalil AA, Poulton J, Castillejo-Lopez C, Egger-Adam D, Wodarz A, Deng W-M, and S Baumgartner. (2006) Perlecan and Dystroglycan act at the basal side of the Drosophila follicular epithelium to maintain epithelial organization. Development. 133:3805-3815
- Poulton J and W-M Deng. (2007) Cell–cell communication and axis specification in the Drosophila oocyte. Developmental Biology. 311(1):1-10 Yu J*, Poulton J*, Huang YC, and W-M Deng. (2008) The Hippo pathway promotes Notch signaling in regulation of cell differentiation, proliferation, and oocyte polarity. PLoS One. 3:e1761 *denotes equal contribution
- Sagona AP, Nezis IP, Pedersen NM, Liestøl K, Poulton J, Rusten TE, Skotheim RI, Raiborg C, Stenmark H. (2010) PtdIns(3)P controls cytokinesis through KIF13A-mediated recruitment of FYVE-CENT to the midbody. Nature Cell Biology. 12:362-71.
- Tamori Y, Bialucha CU, Tian AG, Kajita M, Huang YC, Norman M, Harrison N, Poulton J, Ivanovitch K, Disch L, Liu T, Deng WM, Fujita Y. (2010) Involvement of Lgl and Mahjong/VprBP in cell competition. PLoS Biology. 8:e1000422.
- Roberts DM, Pronobis MI, Poulton JS, Waldmann JD, Stephenson EM, Hanna S, Peifer M. (2011) Deconstructing the Beta-Catenin destruction complex: mechanistic roles for the tumor suppressor APC in regulating Wnt signaling. Molecular Biology of the Cell. 22:1845-63.
- Poulton JS, Huang YC, Smith L, Sun J, Leake N, Schleede J, Stevens LM, Deng WM. (2011) The microRNA pathway regulates the temporal pattern of Notch signaling in Drosophila follicle cells. Development. 138:1737-45.
- Roberts,D.M., Pronobis, M.I., Poulton, J.S., Kane,E.G., and Peifer, M. (2012). Regulation of Wnt signaling by the tumor suppressor APC does not require ability to enter the nucleus nor a particular cytoplasmic localization. Molecular Biology of the Cell,23, 2041-2056.
- Roberts,D.M., Pronobis, M.I., Alexandre, K.M., Rogers, G.C., Poulton, J.S., Schneider, D.E., Jung, K.-C., McKay, D.J., and Peifer, M. (2012). Defining components of the ß-catenin destruction complex and exploring its regulation and mechanisms of action during development. PLoS One 7: e31284.
- Gao, L., Shao, L., Higgins, C.D., Poulton, J.S., Peifer, M., Davidson, M.W., Wu, X., Goldstein, B., and Betzig, E. Noninvasive Imaging of Three-Dimensional Dynamics in Thickly Fluorescent Specimens Beyond the Diffraction Limit. Cell 151, 1370-1385.
- Poulton, J.S., Mu, F.W., Roberts, D.M., and Peifer, M. (2013). APC2 and Axin promote mitotic fidelity by facilitating centrosome separation and cytoskeletal regulation. Development 140, 4226-36. [PubMed]
- Poulton, J.S., Cuningham, J.C., and Peifer, M. (2014). Acentrosomal Drosophila epithelial cells exhibit abnormal cell division, leading to cell death and compensatory proliferation. Developmental Cell 30, 731-745. [PubMed]
- Lerit DA, Jordan HA, Poulton JS, Fagerstrom CJ, Galletta BJ, Peifer M, Rusan NM. (2015). Interphase centrosome organization by the PLP-Cnn scaffold is required for centrosome function. J Cell Biol. 210:79-97 [PubMed]
-
Terzo EA, Lyons SM, Poulton JS, Temple BR, Marzluff WF, Duronio RJ. (2015). Distinct self-interaction domains promote Multi Sex Combs accumulation in and formation of the Drosophila histone locus body. Mol Biol Cell. 26:1559-74. [PubMed]
- Lerit DA, Poulton JS. (2016). Centrosomes are multifunctional regulators of genome stability. Chromosome Res. 24:5-17. [PubMed]
- Lo PK, Huang YC, Poulton JS, Leake N, Palmer WH, Vera D, Xie G, Klusza S, Deng WM. (2016) RNA helicase Belle/DDX3 regulates transgene expression in Drosophila. Dev Biol. 412:57-70. [PubMed]
- Poulton, J.S., Cuningham, J.C., and Peifer, M. (2017). Centrosome and Spindle Assembly Checkpoint Loss Leads to Neural Apoptosis and Reduced Brain Size. Journal of Cell Biology 216, 1255-1265. [PUBMED]
- Poulton, J.S. McKay, D. J., and Peifer M. (2019). Centrosome loss triggers a transcriptional program to counter apoptosis-induced oxidative stress. Genetics 212, 187-211. [PUBMED]